Satisfaction Guaranteed
All products are handled with strict quality standards to ensure consistent research-grade excellence.
Secure Ordering
Our checkout is SSL encrypted and completely secure.
Third-party Tested
Our products are verified by independent third party laboratories to meet quality standards.
Batch & Lot Tracking
All product batches and lots are assigned unique identifiers and tied to publicly posted lab reports.
Endless Commitment
We are committed to helping our customers. If you have any questions or concerns, please reach out through our Contact Us page.
TB-500
$74.99
Save 5.01$ (6% Off)
Discount per Quantity
| Quantity | Discount | Price |
|---|---|---|
| 5 - 10 | 5% | $71.24 |
| 11 - 20 | 10% | $67.49 |
| 21+ | 15% | $63.74 |
Rigorous Third-Party Testing
Every batch of our research chemicals and peptides undergoes third-party testing.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes. Kindly review our Terms & Conditions before making a purchase.

Always quality-tested, verified with third party COA’s
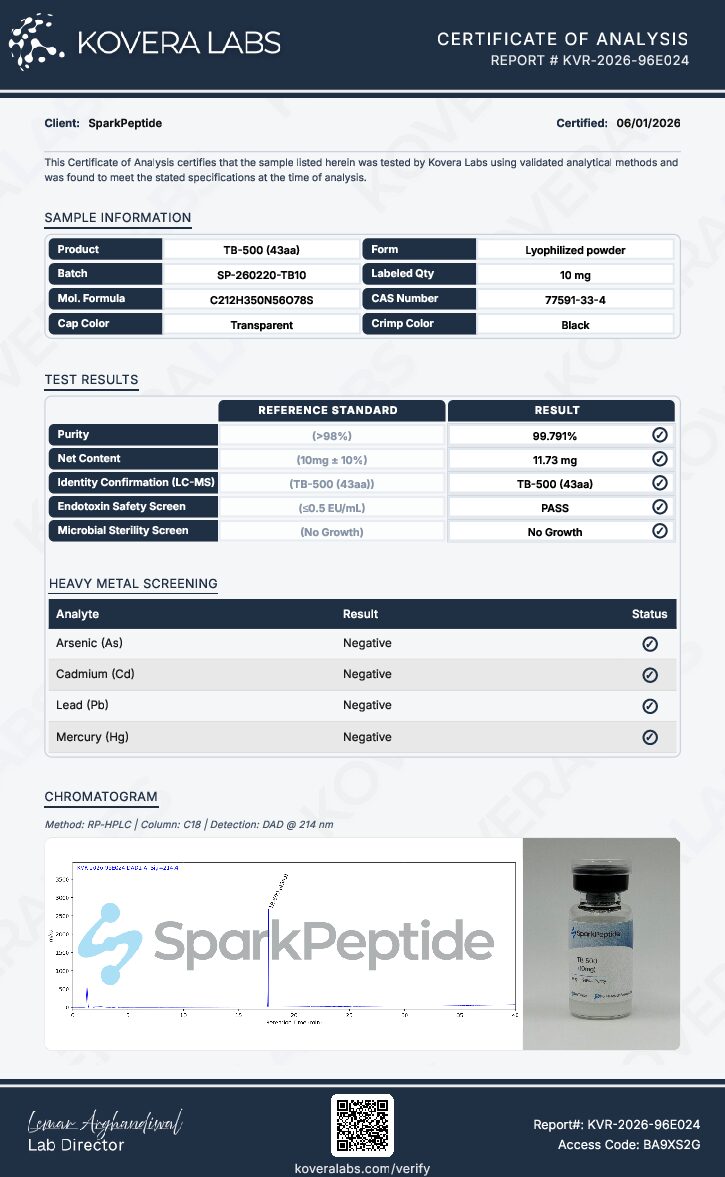
At every step, we prioritize quality by conducting rigorous third-party testing on all our products. These tests focus on five key characteristics- identity, purity, sterility, and endotoxin levels, and heavy metal content-ensuring that each product meets the highest standards of quality with independent third-party Certificates of Analysis (COAS) to verify our commitment to excellence.
Identity Test
Purity Test
Sterility Test
Endotoxin Test
Heavy Metals Test
Identity Test
Purity Test
Sterility Test
Endotoxin Test
Heavy Metals Test
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes.Kindly review our Terms & Conditions before making a purchase.
TB-500 (10mg) Overview
Researchers can buy TB-500 (10mg) for research in experimental systems investigating cellular migration, cytoskeletal dynamics, and tissue repair signaling pathways. TB-500 is a synthetic research peptide derived from the active region of thymosin beta-4 and is widely studied in models of regenerative biology and actin-mediated cellular processes. Spark Peptide supports research reliability through rigorous analytical verification and batch-specific Certificates of Analysis, making the peptide suitable for controlled laboratory investigations such as signaling pathway studies and tissue repair models.Molecular Origin
TB-500 is a synthetic peptide derived from thymosin beta-4, a naturally occurring 43-amino-acid protein found in many mammalian tissues and involved in actin regulation and cellular migration. Thymosin beta-4 plays a role in cytoskeletal organization, angiogenesis, and tissue remodeling processes, making it an important molecule in studies of cellular repair and regenerative biology. The peptide fragment known as TB-500 corresponds to the biologically active region of thymosin beta-4 responsible for many of its actin-binding and cell-motility–related properties. Early research on thymosin peptides began in the 1960s during investigations into thymic hormones and immune regulation, with thymosin beta-4 later identified as a key component influencing cellular movement and structural protein dynamics. TB-500 replicates this functional domain in a smaller synthetic form, maintaining a defined amino acid sequence that enables researchers to isolate and examine specific biological interactions. The peptide is typically produced using solid-phase peptide synthesis (SPPS), a method that sequentially assembles amino acids on a resin support to achieve precise sequence control and high reproducibility. This structural specificity allows TB-500 to serve as a useful model for studying actin-mediated cellular signaling and tissue repair mechanisms.Purity & Quality Standards
TB-500 supplied by Spark Peptide is produced to 99.9%+ purity verified through high-performance liquid chromatography (HPLC) analysis, supporting consistent experimental reproducibility. The rigorous synthesis follows cGMP-certified processes aligned with ISO 9001:2015 quality management standards to ensure controlled production and documentation. More importantly, each batch undergoes Spark Peptide’s 6X Safety Testing protocol, including:- HPLC purity verification
- Mass spectrometry identity confirmation
- Heavy metal screening
- Endotoxin testing
- Bacterial contamination analysis
- Solubility and stability evaluation.
TB-500 Mechanism of Action
TB-500, a synthetic fraction of the naturally occurring protein Thymosin Beta-4, functions as a potent regenerative agent by modulating cellular architecture and initiating targeted repair pathways.Receptor Binding & Primary Signaling
TB-500 does not appear to exert its activity through a single defined membrane receptor. Instead, mechanistic studies indicate that the peptide fragment derived from thymosin beta-4 interacts directly with intracellular actin monomers, influencing cytoskeletal dynamics rather than classical ligand–receptor signaling pathways[1]. The parent protein thymosin beta-4 contains an actin-binding motif that sequesters globular actin (G-actin), regulating actin polymerization and filament assembly within cells. Structural analyses have identified a conserved LKKTET sequence motif responsible for high-affinity interaction with actin monomers, allowing thymosin-derived peptides to influence actin filament turnover and cellular motility processes[2]. Following cellular uptake or diffusion, TB-500–related fragments can modulate the equilibrium between monomeric and filamentous actin pools. This interaction affects cytoskeletal organization, enabling dynamic remodeling of actin filaments that supports processes such as cell migration, intracellular transport, and structural stabilization[1]. Rather than initiating classical GPCR or kinase signaling directly, the peptide’s activity is associated with cytoskeletal regulation that subsequently influences signaling pathways linked to cell adhesion, motility, and structural integrity. Experimental studies using cultured cells and structural binding assays have demonstrated that modulation of actin dynamics can indirectly affect signaling pathways involved in cellular migration and tissue remodeling responses[3].Downstream Biological Cascades
Modulation of actin dynamics by thymosin beta-4–derived peptides such as TB-500 can influence several downstream signaling pathways associated with cellular migration, angiogenesis-related signaling, and cytoskeletal remodeling. Alterations in actin polymerization affect focal adhesion formation and integrin-mediated signaling complexes, which are known to interact with intracellular cascades including PI3K/Akt and MAPK/ERK pathways in experimental cellular systems. These pathways regulate cytoskeletal organization, cell survival signaling, and migration-associated gene expression. In vitro experiments have demonstrated that changes in actin availability can alter transcriptional responses linked to cellular motility and extracellular matrix interaction. For example, actin-regulated signaling has been associated with modulation of transcription factors that coordinate cytoskeletal proteins, adhesion molecules, and growth-related signaling mediators. Animal model studies investigating thymosin beta-4 activity have also reported alterations in angiogenic signaling markers and cytoskeletal regulatory proteins following peptide exposure in controlled experimental conditions. Because cytoskeletal remodeling influences multiple signaling networks, TB-500 is frequently used in laboratory research examining cell migration, wound-healing models, and actin-mediated intracellular signaling. These experimental systems allow researchers to investigate how actin-regulatory peptides contribute to broader biological processes involving tissue structure, vascular growth signaling, and cellular repair mechanisms.Key Scientific Features & Chemical Profile of TB-500
Produced using solid-phase peptide synthesis, TB-500 is a synthetic analogue of the naturally occurring protein Thymosin Beta-4, designed to replicate its core bioactive sequence while providing consistent, well-characterized material for laboratory investigations. Its defined biochemical properties and established interactions with actin polymerization pathways make it a widely utilized compound in experimental models examining tissue repair mechanisms, cytoskeletal dynamics, and cellular migration processes.Molecular Data
| Property | Value |
| Molecular Formula | C212H350N56O78S |
| Molecular Weight | 4963 g/mol |
| Amino Acid Sequence | SDKPDMAEXEKFDKSKLKKXEXQEKNPLPSKEXXEQEKQAGES |
| CAS Number | 77591-33-4 |
| PubChem CID | 45382195 |
| Synonyms | Thymosin Beta 4 THYMOSIN BETA 4 ACETATE |
| Physical Form | Lyophilized white powder |
| Solubility | Soluble in sterile water and DMSO |
| Storage | -4°F (-20°C), desiccated, protected from light |
Analytical Verification
Each batch of TB-500 supplied by Spark Peptides is accompanied by a verified Certificate of Analysis (COA) documenting the compound's comprehensive analytical profile and batch-specific testing data. To ensure researchers receive material of the highest quality, every lot undergoes a rigorous, multi-method verification process prior to release. Chromatographic Purity Assessment High-Performance Liquid Chromatography (HPLC) is employed to determine purity by resolving peptide components within the sample matrix and quantifying their relative abundance. This method provides precise, quantitative confirmation of purity levels at 99.9%+, ensuring that each batch is free from significant impurities or degradation products that could compromise experimental integrity. Molecular Identity Confirmation Molecular identity is independently verified through mass spectrometry (MS), which compares the experimentally observed molecular mass against the theoretical mass derived from TB-500's defined amino acid sequence. This orthogonal approach provides unambiguous confirmation that the correct peptide has been synthesized and isolated. 6X Safety Testing Protocol Beyond chromatographic and spectroscopic analysis, each batch is subject to Spark Peptides' comprehensive 6X Safety Testing protocol, which encompasses the following assessments:- Heavy Metal Screening: Detection and quantification of trace heavy metal contaminants that may interfere with biological assays or compromise researcher safety
- Endotoxin Testing: Evaluation of bacterial endotoxin levels to ensure material is suitable for sensitive cell-based and in vivo experimental applications
- Bacterial Contamination Analysis: Microbiological screening to confirm the absence of viable bacterial organisms within the final product
- Solubility Verification: Confirmation that TB-500 dissolves predictably under defined conditions, supporting consistent reconstitution across experimental protocols
- Stability Assessment: Evaluation of compound integrity under controlled storage conditions to establish reliable shelf-life parameters
Storage, Handling, and Reconstitution
Proper storage and preparation are important for preserving peptide integrity and ensuring consistent experimental results. The following guidelines outline recommended laboratory practices for storing lyophilized TB-500, preparing it for experimental use, and maintaining stability after reconstitution. These procedures help minimize degradation, contamination, and variability during handling and routine laboratory applications.Recommended Storage Conditions
TB-500 should be stored in its lyophilized form at -4°F (-20°C) to maintain long-term stability and structural integrity. Vials should remain tightly sealed and stored in a desiccated environment to minimize exposure to moisture and atmospheric contaminants. Protection from direct light is recommended. Under proper conditions, lyophilized peptides typically maintain stability for extended periods. After reconstitution, the peptide solution should be refrigerated at 36–46°F (2–8°C) and used within an appropriate laboratory timeframe.Reconstitution Protocol
Researchers typically reconstitute TB-500 using bacteriostatic water or another sterile laboratory-grade solvent appropriate for peptide preparation.- Allow the sealed peptide vial to reach room temperature before opening.
- Prepare sterile solvent, commonly bacteriostatic water (see Spark Peptide’s Bacteriostatic Water 10ml product).
- Using a sterile syringe, slowly add the solvent along the inside wall of the vial to minimize foaming.
- Avoid vortexing or vigorous agitation.
- Gently swirl the vial until the lyophilized powder dissolves completely.
- Typical laboratory preparations use solvent volumes between 1–5 mL depending on the desired working concentration.
Handling Precautions
- Handle in a clean, controlled laboratory or sterile preparation environment
- Avoid repeated freeze–thaw cycles that may degrade peptide structure
- Use appropriate personal protective equipment (PPE), including gloves and lab coats
- Follow established laboratory research handling protocols
- Supplied strictly for laboratory research use only; not for human or clinical use
TB-500 Research & Scientific Applications
TB-500, a synthetic fragment derived from thymosin beta-4, is widely investigated in laboratory research examining cytoskeletal regulation, cellular migration, and tissue remodeling processes. Published findings indicate that thymosin beta-4–derived peptides influence actin dynamics and signaling networks associated with cellular motility, angiogenic signaling, and structural tissue organization[6]. As a result, TB-500 is commonly used in experimental systems exploring actin-mediated signaling pathways and regenerative biology mechanisms.Preclinical & Diagnostic Research
In vitro research has extensively examined thymosin beta-4 and related peptide fragments such as TB-500 in controlled cellular systems. Studies have demonstrated that the peptide interacts with globular actin (G-actin), regulating the equilibrium between monomeric and filamentous actin pools that determine cytoskeletal organization. These mechanisms are commonly investigated using cultured fibroblasts, endothelial cells, and epithelial cell lines to measure endpoints such as actin polymerization dynamics, cytoskeletal protein distribution, and cellular migration behavior(3). Cell-based experiments have also explored how thymosin beta-4–derived peptides influence intracellular signaling cascades linked to cytoskeletal remodeling. Published findings indicate activation of pathways associated with PI3K/Akt and MAPK/ERK signaling in experimental cell systems, which are known to regulate cellular motility, adhesion, and survival-related gene expression. These responses are typically measured using molecular assays evaluating phosphorylation states of signaling proteins, transcription factor activation, and downstream gene expression changes(3). Additional in-vitro investigations have focused on endothelial cell models to study vascular signaling pathways. Experimental observations have shown that thymosin beta-4 peptides can influence markers associated with angiogenic signaling and endothelial migration. Laboratory assays examining endothelial tube formation, cytoskeletal reorganization, and extracellular matrix interactions have been used to investigate these responses(7). Researchers have also used thymosin beta-4 fragments to examine cytoskeleton-dependent intracellular transport processes. Because actin filaments serve as scaffolding for intracellular trafficking, TB-500 has been studied in experimental systems investigating vesicle transport, cytoskeletal stabilization, and structural protein assembly in cultured cell models.Animal Model Observations
Animal studies involving thymosin beta-4 and related peptide fragments have provided insights into the broader physiological roles of actin-regulating peptides. Rodent models are frequently used to investigate how modulation of actin dynamics influences tissue remodeling and vascular signaling pathways. Experimental findings suggest that thymosin-derived peptides can alter the expression of biomarkers associated with endothelial cell migration, extracellular matrix regulation, and cytoskeletal protein turnover(8). In murine injury models, researchers have observed changes in molecular markers linked to angiogenesis and cytoskeletal remodeling within regenerating tissues. Studies examining vascular and skeletal muscle systems reported increased expression of endothelial migration markers and proteins involved in actin filament organization following exposure to thymosin beta-4–derived peptides. These endpoints are typically measured using immunohistochemistry, protein expression analysis, and gene transcription profiling(6). Additional experimental work in animal models has explored how thymosin beta-4 signaling influences inflammatory signaling networks and tissue structural integrity. Published findings have reported modulation of cytokine signaling markers, extracellular matrix proteins, and cellular migration factors in injured tissues. These observations suggest that thymosin-derived peptides interact with multiple signaling networks regulating tissue remodeling, vascular growth signaling, and cytoskeletal organization in preclinical experimental systems(7).Cytoskeletal Dynamics & Cell Migration Studies
A major research focus involving TB-500 centers on the regulation of cytoskeletal dynamics and cellular motility. Thymosin beta-4–derived peptides are widely used in experimental systems designed to study how actin-binding proteins regulate cell movement and structural reorganization. Because actin polymerization is fundamental to processes such as wound closure, immune cell migration, and tissue remodeling, TB-500 provides a useful molecular tool for investigating actin-mediated signaling pathways(3). Experimental models frequently examine changes in actin filament formation, focal adhesion turnover, and cytoskeletal protein interactions following exposure to thymosin-derived peptides. Quantifiable endpoints in these systems include actin filament density, cellular migration rates, and expression of cytoskeleton-associated regulatory proteins. These studies contribute to a broader understanding of how actin-binding peptides influence cellular architecture and dynamic signaling networks involved in structural tissue biology.TB-500 Comparative Analysis
TB-500 is frequently compared with other peptides studied in tissue remodeling and cellular migration research, particularly thymosin beta-4 (Tβ4) and BPC-157. Unlike receptor-binding peptides that activate specific membrane receptors, TB-500 functions primarily through interaction with actin monomers, influencing cytoskeletal organization and cell motility mechanisms. TB-500 represents a shorter synthetic fragment derived from the active region of thymosin beta-4, allowing researchers to investigate actin-regulatory signaling without the full-length 43–amino acid protein structure. Comparative studies indicate that full-length thymosin beta-4 exhibits broader biological activity due to its complete structural sequence, which participates in multiple cytoskeletal and signaling interactions. TB-500 isolates the actin-binding functional domain, enabling more targeted examination of actin polymerization and cellular migration pathways in experimental systems. In contrast, BPC-157, a peptide derived from a gastric protein fragment, is investigated primarily for signaling pathways associated with nitric oxide regulation and angiogenic signaling in laboratory models. Experimental models suggest that while TB-500 and thymosin beta-4 primarily influence cytoskeletal remodeling through actin-binding mechanisms, BPC-157 is often studied in signaling networks involving vascular growth factors, nitric oxide signaling, and extracellular matrix regulation. As a result, TB-500 is commonly used in laboratory systems examining cytoskeletal dynamics and cell migration processes, whereas BPC-157 research frequently focuses on endothelial signaling and vascular-related pathways.| Parameter | TB-500 | Thymosin Beta-4 | BPC-157 |
| Half-life | Moderate peptide stability in experimental systems | Short-lived endogenous peptide | Reported stability in peptide research models |
| Receptor Selectivity | No defined membrane receptor; actin-binding interaction | Actin-binding protein; multiple intracellular interactions | Associated with signaling pathways involving nitric oxide and growth factors |
| Primary Mechanism | Regulation of actin polymerization and cytoskeletal remodeling | Broad cytoskeletal regulation and signaling interactions | Modulation of angiogenic and nitric oxide signaling pathways |
| Research Applications | Cytoskeletal dynamics, cell migration models, tissue remodeling studies | Actin regulation, angiogenesis signaling, tissue remodeling research | Vascular signaling studies, endothelial cell models, extracellular matrix signaling |
Peer-Reviewed Research & Citations
- Cassimeris, L., Safer, D., Nachmias, V.T., and Zigmond, S.H. "Thymosin Beta 4 Sequesters the Majority of G-Actin in Resting Human Polymorphonuclear Leukocytes." Journal of Cell Biology, vol. 119, no. 5, pp. 1261–1270, 1992. PMID: 1447300 / DOI: 10.1083/jcb.119.5.1261
- Sun, J., Zhong, X., Fu, X., Miller, H., Lee, P., Yu, B., and Liu, C. "The Actin Regulators Involved in the Function and Related Diseases of Lymphocytes." Frontiers in Immunology, vol. 13, pp. 799309, 2022. DOI: 10.3389/fimmu.2022.799309
- Ying, Y., Tao, N., Zhang, F., Wen, X., Zhou, M., and Gao, J. "Thymosin β4 Regulates the Differentiation of Thymocytes by Controlling the Cytoskeletal Rearrangement and Mitochondrial Transfer of Thymus Epithelial Cells." International Journal of Molecular Sciences, vol. 25, no. 2, 1088, 2024. PMID: 38256161 / DOI: 10.3390/ijms25021088
- Xing, Y., Ye, Y., and Li, Y. "Progress on the Function and Application of Thymosin β4." Frontiers in Endocrinology, vol. 12, pp. 767785, 2021. DOI: 10.3389/fendo.2021.767785
- Su, L., Kong, X., Loo, S., Gao, Y., Liu, B., Su, X., Dalan, R., Ma, J., and Ye, L. "Thymosin Beta-4 Improves Endothelial Function and Reparative Potency of Diabetic Endothelial Cells Differentiated from Patient Induced Pluripotent Stem Cells." Stem Cell Research & Therapy, vol. 13, no. 1, 13, 2022. PMID: 35012642 / DOI: 10.1186/s13287-021-02687-x
- Maar, K., Hetenyi, R., Maar, S., Faskerti, G., Hanna, D., Lippai, B., Takatsy, A., and Bock-Marquette, I. "Utilizing Developmentally Essential Secreted Peptides Such as Thymosin Beta-4 to Remind the Adult Organs of Their Embryonic State—New Directions in Anti-Aging Regenerative Therapies." Cells, vol. 10, no. 6, 1343, 2021. PMID: 34071596 / DOI: 10.3390/cells10061343
Certificate of Analysis & Lab Reports
Each batch of TB-500 supplied by Spark Peptide is accompanied by analytical documentation generated by independent third-party laboratories. These reports verify compound identity, confirm purity levels, and document safety screening results. The documentation is part of Spark Peptide’s 6X Safety Testing protocol, providing researchers with batch-specific analytical verification to support reproducibility and laboratory quality assurance.Certificate of Analysis (COA)
The Certificate of Analysis documents the analytical results for the specific manufacturing lot supplied to researchers. It confirms the compound’s molecular identity, verifies purity measurements obtained through validated analytical methods, and records key safety parameters. The COA also provides batch traceability information, including lot identification and testing documentation generated during quality verification.HPLC Analysis Report
High-Performance Liquid Chromatography (HPLC) is used to analyze the composition of the peptide sample by separating individual components within the mixture. This technique allows analytical laboratories to identify potential impurities and quantify the relative concentration of the target peptide. The resulting chromatogram provides a visual representation of compound purity based on peak separation and signal intensity.Mass Spectrometry Report
Mass spectrometry is used to confirm molecular identity by measuring the mass-to-charge ratio (m/z) of ionized peptide fragments. The observed mass spectrum is compared with the theoretical molecular mass derived from the peptide’s amino acid sequence. This comparison verifies that the detected molecular profile corresponds to the expected structure of TB-500.Additional Safety Screening
In addition to HPLC purity verification and mass spectrometry identity confirmation, TB-500 undergoes additional analytical screening as part of Spark Peptide’s 6X Safety Testing protocol. These procedures include heavy metals screening for lead, mercury, arsenic, and cadmium; endotoxin testing using the Limulus Amebocyte Lysate (LAL) assay; and bacterial contamination analysis. Together, these tests help verify chemical safety and laboratory suitability. Complete analytical reports are available upon request or through the Spark Peptide Tests & Safety page.6X Safety Testing
While most suppliers verify purity alone, every SparkPeptide batch passes six independent quality and safety screenings before reaching your laboratory.| # | Test | What It Confirms |
| 1 | HPLC Purity Analysis | Peptide purity at 99.9%+ via reverse-phase chromatography |
| 2 | Mass Spectrometry | Correct molecular identity (observed vs. expected mass) |
| 3 | Heavy Metal Screening | Below detectable limits for lead, mercury, arsenic, cadmium |
| 4 | Endotoxin Testing | Bacterial endotoxin levels within safe research thresholds (LAL assay) |
| 5 | Bacterial Contamination | No microbial growth detected in culture testing |
| 6 | Solubility & Stability | Proper reconstitution behavior and structural integrity confirmed |
Legal Disclaimer
For Laboratory Research Use Only. All products sold by Spark Peptide are strictly intended for laboratory research use only. These materials are not for human consumption and are not intended for medical, veterinary, diagnostic, or household use of any kind. Spark Peptide operates solely as a research chemical supplier. We are not a compounding pharmacy and do not operate as a compounding facility as defined under Section 503A of the Federal Food, Drug, and Cosmetic Act. Additionally, Spark Peptide is not registered as an outsourcing facility under Section 503B of the Act. By purchasing from our site, you agree to use our products exclusively for lawful laboratory research purposes. Any misuse is strictly prohibited.Product FAQ for Researchers
What purity level does Spark Peptide’s TB-500 achieve?
Spark Peptide’s TB-500 is produced to 99.9%+ purity verified via High-Performance Liquid Chromatography (HPLC). Each manufacturing batch undergoes the company’s 6X Safety Testing protocol, which includes HPLC purity analysis, mass spectrometry identity confirmation, heavy metal screening, endotoxin testing, bacterial contamination analysis, and solubility/stability verification. Batch-specific analytical results are documented in the accompanying Certificate of Analysis (COA).How should TB-500 be reconstituted for laboratory use?
TB-500 is typically reconstituted using sterile bacteriostatic water or another laboratory-grade solvent appropriate for peptide preparation. Researchers should allow the vial to reach room temperature, then slowly introduce the solvent along the vial wall and gently swirl until fully dissolved and avoid vigorous agitation. For convenience, Spark Peptide offers a compatible Bacteriostatic Water 10ml product commonly used in peptide preparation.What is the shelf life of TB-500?
When stored properly in its lyophilized form at -4°F (-20°C) and protected from light and moisture, TB-500 can remain stable for extended periods under standard laboratory storage conditions. After reconstitution, the peptide solution should be refrigerated at 36–46°F (2–8°C) and typically used within a short-term research timeframe to maintain structural integrity.Does TB-500 include a Certificate of Analysis?
Yes, every TB-500 batch supplied by Spark Peptide is accompanied by a batch-specific Certificate of Analysis (COA). The COA documents the analytical testing results for that production lot, including identity confirmation, purity verification, and safety screening parameters. Researchers can view or download the COA directly from the product page or request additional documentation if required.How is TB-500 shipped?
Spark Peptide packages TB-500 in protective laboratory-grade vials designed to help limit temperature fluctuations during transit. Lyophilized peptides generally maintain stability under typical ambient shipping conditions. Orders are processed promptly, with our dedicated support team helping ensure reliable delivery to individual researchers and institutional buyers in the U.S.How does TB-500 differ from full-length thymosin beta-4?
TB-500 represents a synthetic fragment derived from the active region of thymosin beta-4, a naturally occurring actin-binding protein. While full-length thymosin beta-4 contains 43 amino acids and participates in multiple cellular signaling processes, TB-500 isolates a functional sequence associated with actin regulation and cytoskeletal dynamics. This allows researchers to study actin-mediated cellular migration and structural signaling pathways in controlled experimental systems.| Property | Detail |
|---|---|
| Name | TB-500 (synthetic thymosin beta-4 analogue) |
| Classification | Synthetic peptide analogue modeled on thymosin beta-4 (Tβ4) activity |
| Sequence | Ac-Ser-Asp-Lys-Pro-Asp-Met-Ala-Glu-Ile-Glu-Lys-Phe-Asp-Lys-Ser-Lys-Leu-Lys-Lys-Thr-Glu-Thr-Gln-Glu-Lys-Asn-Pro-Leu-Pro-Ser-Lys-Glu-Thr-Ile-Glu-Gln-Glu-Lys-Gln-Ala-Gly-Glu-Ser-OH |
| Molecular Formula | C₂₁₂H₃₅₀N₅₆O₇₈S |
| Molecular Weight | ~5 kDa (may vary slightly depending on synthesis and analogue structure) |
| Peptide Length | 43 amino acids |
| Primary Research Pathways | Actin sequestration and cytoskeletal regulation; cellular migration; angiogenesis signaling; inflammatory modulation; cytoprotection and extracellular matrix remodeling |
| Format | Lyophilized powder supplied in sealed sterile glass research vials |
| Purity | ≥99%, verified by lot-specific third-party Certificate of Analysis (COA) |
| Solubility | Soluble in bacteriostatic water or approved research-grade buffers; introduce solvent gently to avoid foaming |
| Storage (Lyophilized) | –20°C to –80°C (–4°F to –112°F), protected from light and moisture |
| Storage (Reconstituted) | 2–8°C (36–46°F) for short-term working solutions; aliquot into single-use volumes and store frozen for long-term use |
| Handling Notes | Avoid vigorous agitation during reconstitution; minimize freeze–thaw cycles; follow standard laboratory biosafety protocols |
| Batch | EP-250421-TB05 |
| Research Designation | For research use only — not approved for human or veterinary use, clinical administration, or therapeutic application |
| Supplier | Spark Peptide |
Related products
-
Peptides
GHK-Cu
$54.99 – $79.99Price range: $54.99 through $79.99 Purchase & earn 80 points!Select options -
Peptides
BPC-157
$39.99 – $64.99Price range: $39.99 through $64.99 Purchase & earn 65 points!Select options