Satisfaction Guaranteed
All products are handled with strict quality standards to ensure consistent research-grade excellence.
Secure Ordering
Our checkout is SSL encrypted and completely secure.
Third-party Tested
Our products are verified by independent third party laboratories to meet quality standards.
Batch & Lot Tracking
All product batches and lots are assigned unique identifiers and tied to publicly posted lab reports.
Endless Commitment
We are committed to helping our customers. If you have any questions or concerns, please reach out through our Contact Us page.
BPC-157
$39.99 – $64.99Price range: $39.99 through $64.99
Save 5.01$ (7% Off)
Discount per Quantity
| Quantity | Discount | Price |
|---|---|---|
| 5 - 10 | 5% | $61.74 |
| 11 - 20 | 10% | $58.49 |
| 21+ | 15% | $55.24 |
Rigorous Third-Party Testing
Every batch of our research chemicals and peptides undergoes third-party testing.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes. Kindly review our Terms & Conditions before making a purchase.

Always quality-tested, verified with third party COA’s
At every step, we prioritize quality by conducting rigorous third-party testing on all our products. These tests focus on five key characteristics- identity, purity, sterility, and endotoxin levels, and heavy metal content-ensuring that each product meets the highest standards of quality with independent third-party Certificates of Analysis (COAS) to verify our commitment to excellence.
Identity Test
Purity Test
Sterility Test
Endotoxin Test
Heavy Metals Test
Identity Test
Purity Test
Sterility Test
Endotoxin Test
Heavy Metals Test
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes.Kindly review our Terms & Conditions before making a purchase.
BPC-157 Overview
Independent researchers and research institutions often buy BPC-157 from Spark Peptides for use in experiments involving nitric oxide signaling, angiogenic pathways, and cytoprotective cellular responses. That’s because Spark Peptide provides ultra-pure BPC-157 for sale tested with rigorous analytical verification methods by our third-party testing partners, with batch-specific certificates of analysis to match. BPC-157 is frequently investigated in receptor pathway studies, cellular signaling assays, and laboratory models examining peptide-mediated regulation of tissue and vascular biology.Molecular Origin
BPC-157 (Body Protection Compound-157) is a synthetic pentadecapeptide derived from a naturally occurring protective protein sequence originally isolated from human gastric juice[1]. The peptide belongs to a class of cytoprotective peptide fragments studied for their involvement in cellular repair processes and regulatory signaling networks associated with gastrointestinal physiology. Early biochemical investigations during the 1980s and 1990s identified short peptide fragments within gastric protective proteins that appeared to influence mucosal defense mechanisms and vascular signaling pathways. BPC-157 represents a stable fragment of these proteins, consisting of a defined fifteen–amino-acid sequence that retains the biologically active region of the parent compound. Structurally, the peptide is characterized by a stable linear amino acid sequence capable of interacting with signaling systems related to nitric oxide pathways, endothelial function, and cellular stress responses. Synthetic production of BPC-157 relies on solid-phase peptide synthesis (SPPS), which enables precise assembly of the peptide sequence through sequential amino acid coupling. The peptide is then cleaved from the resin and protective side-chain groups are removed, a critical stage that transitions the compound from a solid-bound state to a crude liquid form. This crude material then undergoes a rigorous purification sequence, typically utilizing preparative HPLC, to isolate the target BPC-157 sequence from any residual reagents or synthesis byproducts. This controlled synthesis process allows researchers to obtain highly defined peptide material suitable for investigating receptor interactions, intracellular signaling cascades, and peptide-mediated regulatory mechanisms in experimental models.Purity & Quality Standards
BPC-157 supplied by Spark Peptide is produced to 99.9%+ purity verified through high-performance liquid chromatography (HPLC), supporting consistent analytical quality for laboratory research applications. The company’s manufacturing follows cGMP-certified processes and aligns with ISO 9001:2015 quality management standards to maintain rigorous production oversight. Plus, each batch undergoes Spark Peptide’s 6X Safety Testing protocol, including HPLC purity verification, mass spectrometry identity confirmation, heavy metal screening, endotoxin testing, bacterial contamination analysis, and solubility and stability testing. Verified Certificates of Analysis (COAs) accompany every lot to certify this quality and purity. To maintain stability, lyophilized peptides are packaged using protective materials designed to limit temperature fluctuations during shipping, allowing it to remain stable during standard transit conditions.BPC 157 Mechanism of Action
BPC-157 is widely studied in experimental systems investigating peptide-mediated regulation of vascular signaling, nitric-oxide–associated pathways, and cellular stress responses. Research suggests that the peptide interacts with multiple intracellular signaling networks rather than a single classical receptor target. The following sections summarize primary molecular interactions and downstream signaling events observed in laboratory models.Receptor Binding & Primary Signaling
BPC-157 is considered a signaling peptide fragment that influences several regulatory pathways associated with nitric oxide (NO) signaling and vascular endothelial responses[1]. Experimental studies suggest that the peptide modulates nitric oxide synthase activity and interacts with endothelial regulatory systems responsible for maintaining vascular homeostasis. Rather than binding to a single defined receptor, BPC-157 appears to influence multiple signaling networks through interactions with endothelial cell signaling intermediates and NO-related pathways. Investigations in endothelial cell models indicate that BPC-157 can affect nitric oxide production through modulation of endothelial nitric oxide synthase (eNOS) activity. Nitric oxide signaling plays a central role in vascular tone regulation, cellular migration, and angiogenic signaling cascades. Experimental findings suggest that BPC-157 influences this pathway by stabilizing interactions between nitric oxide regulatory proteins and intracellular signaling mediators. Structural characteristics of the peptide may allow it to interact with regulatory components involved in the VEGF signaling system. Vascular endothelial growth factor receptors belong to the receptor tyrosine kinase family and initiate downstream signaling cascades associated with angiogenesis and endothelial cell migration. Experimental models suggest that BPC-157 may indirectly influence VEGF-related signaling responses and cellular migration pathways[2]. Through these interactions, the peptide has been observed to activate intracellular signaling processes that include nitric-oxide–related pathways and regulatory mechanisms associated with endothelial and cytoprotective cellular responses.Downstream Biological Cascades
Following its interaction with endothelial and nitric-oxide–associated signaling pathways, BPC-157 has been reported to influence several intracellular cascades linked to cellular stress responses, cytoskeletal organization, and transcriptional regulation. Experimental studies indicate that the peptide may activate signaling intermediates associated with the PI3K/Akt and MAPK/ERK pathways, both of which are central regulators of cellular survival, migration, and metabolic adaptation[1]. Activation of the PI3K/Akt pathway has been observed in several cellular assays examining endothelial signaling responses. This pathway regulates numerous cellular processes including gene transcription, cytoskeletal organization, and protein synthesis. Experimental findings suggest that BPC-157 may influence Akt phosphorylation and related signaling events in endothelial and epithelial cell systems. The MAPK/ERK pathway is another signaling cascade frequently examined in BPC-157 research. Activation of ERK kinases plays a key role in regulating gene expression and cellular proliferation responses. Experimental observations have shown changes in ERK-related signaling markers following exposure to the peptide in cultured cellular models. These signaling cascades may influence transcriptional activity of regulatory proteins involved in angiogenic and cytoprotective responses. Studies using cellular assays and animal models suggest measurable changes in gene expression patterns associated with extracellular matrix regulation, inflammatory signaling pathways, and cellular adaptation processes.Key Scientific Features & Chemical Profile of BPC-157
BPC-157 is a synthetic pentadecapeptide derived from a naturally occurring protective protein found in gastric juice, so its chemical profile reflects both the structural precision of its fifteen-amino-acid sequence and the biological activity associated with its parent compound.Molecular Data
| Property | Value |
| Molecular Formula | C62H98N16O22 |
| Molecular Weight | 1419.5 g/mol |
| Amino Acid Sequence | GEPPPGKPADDAGLV |
| CAS Number | 137525-51-0 |
| PubChem CID | 9941957 |
| Synonyms | Bpc 157, Body Protective Compound-157, Pentadecapeptide BPC-157 |
| Physical Form | Lyophilized white powder |
| Solubility | Soluble in water or DMSO |
| Storage | -20°C, desiccated, protected from light |
Analytical Verification
Each batch of BPC-157 supplied by Spark Peptide is accompanied by a Certificate of Analysis (CoA) to serve as a comprehensive record of identity confirmation, purity verification, and batch-specific analytical data. Peptide purity is assessed using High-Performance Liquid Chromatography (HPLC), a gold-standard analytical technique that separates, identifies, and quantifies individual components within the sample matrix. This method confirms purity exceeding 99.9%, effectively detecting and characterizing any residual impurities, synthesis byproducts, or degradation products that could compromise experimental integrity. Molecular identity is independently verified through mass spectrometry, which determines the precise molecular mass of the peptide and compares it against the theoretical mass predicted by the target amino acid sequence. This orthogonal confirmation step ensures that the correct peptide has been synthesized with the intended primary structure, which is a critical safeguard against sequence errors or post-synthetic modifications. Beyond purity and identity, each batch undergoes Spark Peptide's rigorous 6X Safety Testing protocol, encompassing six additional layers of analytical screening:- Heavy metals screening: detects trace elemental contaminants, including lead, mercury, arsenic, and cadmium
- Endotoxin testing: quantifies bacterial lipopolysaccharide (LPS) contamination, a particularly important parameter given that endotoxins are known to trigger inflammatory responses and confound in vitro results
- Bacterial contamination analysis: confirms microbiological purity to rule out viable bacterial presence
- Solubility verification: validates that the peptide dissolves predictably under defined conditions, ensuring consistent reconstitution and accurate dosing across experiments
- Stability assessment: evaluates the peptide's integrity under recommended storage conditions, which also confirms that potency and structural fidelity are preserved
Storage, Handling, and Reconstitution
Proper storage and handling are essential for preserving peptide stability, maintaining structural integrity, and ensuring analytical reliability throughout the course of laboratory use.Recommended Storage Conditions
Lyophilized BPC-157 should be stored at -20°C (-4°F) in its original sealed vial under dry, desiccated conditions, away from light and moisture. Exposure to humidity or photodegradation can accelerate structural breakdown and compromise purity, so controlled storage conditions are critical for maintaining batch integrity. Under these recommended conditions, lyophilized BPC-157 typically remains stable for up to 24 months from the date of manufacture. Following reconstitution, the resulting peptide solution should be transferred to refrigerated storage at 2–8°C (36–46°F). Reconstituted solutions have a reduced stability window compared to lyophilized material and should be used within standard laboratory stability timeframes to avoid potency loss or degradation.Reconstitution Protocol
Careful reconstitution technique is critical for preserving peptide integrity and achieving a consistent, well-characterized working solution.- Remove the vial from frozen storage and allow it to equilibrate to room temperature before opening, preventing condensation from introducing moisture into the lyophilized material.
- Prepare sterile bacteriostatic water as the reconstitution solvent. We recommend that you buy bacteriostatic water from Spark Peptide for guaranteed compatibility and sterility.
- Draw the desired volume of solvent and introduce it slowly along the interior wall of the vial rather than directly onto the lyophilized cake, minimizing foaming and mechanical disruption.
- A typical laboratory preparation uses approximately 1–3 mL of solvent, adjusted according to the target working concentration required for the specific experimental application.
- Do not vortex. Gently swirl or rotate the vial until the peptide is fully dissolved. Aggressive agitation can introduce air bubbles, promote aggregation, or cause structural degradation.
- Visually inspect the final solution; it should appear clear and colorless with no visible particulates. Turbidity or precipitate may indicate incomplete dissolution or compromised peptide integrity.
- Transfer the reconstituted solution to refrigerated storage at 2–8°C (36–46°F) immediately following preparation.
Handling Precautions
Adherence to standard laboratory safety and handling practices is required when working with this material.- All procedures should be conducted in a clean or sterile laboratory environment to prevent introduction of contaminants that could affect sample quality or experimental outcomes
- Repeated freeze–thaw cycles should be strictly avoided, as thermal cycling accelerates peptide degradation and reduces solution stability over time
- Appropriate personal protective equipment (PPE), including nitrile gloves and a laboratory coat, should be worn throughout all handling procedures
- Researchers should follow institutional biosafety protocols and applicable regulatory guidelines governing the handling of research-grade peptide compounds
- BPC-157 supplied by Spark Peptide is intended strictly for in vitro and laboratory research applications and is not approved for human or veterinary use
BPC-157 Research & Scientific Applications
BPC-157 is a synthetic pentadecapeptide that has been extensively investigated in preclinical laboratory research for its role in vascular signaling, cellular stress responses, angiogenesis, and peptide-mediated cytoprotective mechanisms. Originally isolated from gastric juice, the peptide has become a subject of significant scientific interest due to its pleiotropic activity across multiple interconnected biological pathways. All findings referenced below derive from preclinical in vitro and animal model research; BPC-157 has not been approved for human use, and large-scale clinical trials have not yet been conducted.Preclinical & In Vitro Research
One of the most extensively characterized mechanisms involves BPC-157's interaction with the nitric oxide (NO) signaling system. Published preclinical work has demonstrated that BPC-157 modulates nitric oxide production through the Src-Caveolin-1-eNOS pathway, whereby the peptide promotes phosphorylation of Src and Caveolin-1, disrupting the inhibitory Cav-1/eNOS complex and enabling sustained eNOS activation and nitric oxide synthesis[3]. BPC-157 activates both VEGF-dependent signaling (via the VEGFR2–PI3K–Akt–eNOS axis) and VEGF-independent signaling via the Src–caveolin-1–eNOS pathway, with both converging on nitric oxide production to support angiogenesis, vasodilation, and vascular stability[2]. At the kinase signaling level, BPC-157 has been shown to activate two primary downstream cascades: the focal adhesion kinase (FAK)–extracellular signal-regulated kinase (ERK) pathway and the PI3K–Akt pathway, both of which are initiated through Src family kinases. Pharmacological inhibition of these kinases has been shown to abolish BPC-157-induced angiogenesis and endothelial cell migration in vitro, underscoring their mechanistic relevance[3]. Investigations in fibroblast culture systems have further examined the peptide's influence on extracellular matrix organization. Preclinical studies indicate that BPC-157 influences fibroblast activity and collagen fiber production, with observed effects on cell migration and extracellular matrix reorganization consistent with a role in coordinating the cellular environment necessary for tissue regeneration[1]. At the transcriptional level, gene expression studies have identified upregulation of VEGFR2, Nos3, Nos1, Akt1, and Src (genes associated with angiogenesis, nitric oxide synthesis, cell survival, and growth signaling) alongside downregulation of Nos2, which produces inflammatory quantities of nitric oxide and may contribute to tissue damage if overexpressed[1]. Additionally, BPC-157 has been shown to upregulate cytoprotective factors including heme oxygenase-1 (HO-1) and heat shock proteins, with observed effects on mitochondrial integrity and oxidative stress reduction in cellular systems[2].Animal Model Observations
Rodent models have provided the primary body of evidence for BPC-157's in vivo signaling activity. In experimental models of vascular injury, BPC-157 has been shown to markedly promote VEGFR2 expression and angiogenesis in ischemic tissue, with accelerated blood flow recovery attributed to new vessel formation rather than changes in systemic pressure[3][4]. Animal studies have reported measurable effects on musculoskeletal and neuromuscular systems, with BPC-157 demonstrating pro-healing activity particularly in poorly vascularized tissues such as tendons and myotendinous junctions, where angiogenic support is a limiting factor in repair[2]. Inflammatory signaling has also been examined in rodent tissue models. In experimental models of vascular injury, BPC-157 has been associated with restoration of microcirculation and modulation of pathological thrombosis, with findings suggesting the peptide affects not only tissue cells but also the broader vascular environment required for regeneration[2]. Collectively, these experimental observations have contributed to a growing body of preclinical literature examining BPC-157's role in vascular signaling regulation, angiogenic pathway modulation, and cytoprotective cellular mechanisms, while highlighting the need for further research to determine the translational relevance of these findings in human systems.BPC-157 Comparative Analysis
BPC-157 differs from several other peptides investigated in tissue-repair research because it represents a stable fragment of a gastric cytoprotective protein rather than a fragment derived from thymosin or extracellular matrix proteins. Comparative studies frequently examine BPC-157 alongside TB-500 and GHK-Cu, two peptides widely investigated in cellular migration and extracellular matrix research. TB-500, a synthetic fragment derived from thymosin β4, primarily functions through actin-binding interactions that regulate cytoskeletal dynamics and cellular motility. GHK-Cu is a copper-binding tripeptide known to influence extracellular matrix gene expression and metalloproteinase activity. In contrast, experimental models suggest that BPC-157 is associated with nitric oxide signaling pathways and endothelial regulatory mechanisms. These mechanistic differences influence how each peptide is used in experimental research. BPC-157 is commonly investigated in vascular signaling and nitric-oxide–associated pathway studies, whereas TB-500 research focuses on cytoskeletal organization and GHK-Cu research often examines extracellular matrix gene regulation.| Parameter | BPC-157 | TB-500 | GHK-Cu |
| Half-life | Stable peptide fragment | Short thymosin fragment | Small copper-binding tripeptide |
| Receptor Selectivity | NO-associated endothelial pathways | Actin interaction | Integrin & ECM signaling |
| Primary Mechanism | Nitric-oxide signaling modulation | Cytoskeletal regulation | Copper-mediated gene signaling |
| Research Applications | Vascular signaling studies | Cell migration assays | ECM remodeling research |
Peer-Reviewed Research & Citations
- Chang, C.H., Tsai, W.C., Hsu, Y.H., and Pang, J.H. "Pentadecapeptide BPC 157 Enhances the Growth Hormone Receptor Expression in Tendon Fibroblasts." Molecules, vol. 19, no. 11, pp. 19066–19077, 2014. PMID: 25415472 / DOI: 10.3390/molecules191119066
- McGuire, F.P., Martinez, R., Lenz, A., Skinner, L., and Cushman, D.M. "Regeneration or Risk? A Narrative Review of BPC-157 for Musculoskeletal Healing." Current Reviews in Musculoskeletal Medicine, vol. 18, no. 12, pp. 611–619, 2025. PMID: 40789979 / DOI: 10.1007/s12178-025-09990-7
- Hsieh, M.J., Lee, C.H., and Chueh, H.Y. et al. "Modulatory Effects of BPC 157 on Vasomotor Tone and the Activation of Src-Caveolin-1-Endothelial Nitric Oxide Synthase Pathway." Scientific Reports, vol. 10, no. 17078, 2020. DOI: 10.1038/s41598-020-74022-y
- Hsieh, M.J., Liu, H.T., Wang, C.N., Huang, H.Y., Lin, Y., Ko, Y.S., Wang, J.S., Chang, V.H., and Pang, J.S. "Therapeutic Potential of Pro-Angiogenic BPC157 is Associated with VEGFR2 Activation and Up-Regulation." Journal of Molecular Medicine, vol. 95, no. 3, pp. 323–333, 2017. PMID: 27847966 / DOI: 10.1007/s00109-016-1488-y
Certificate of Analysis & Lab Reports
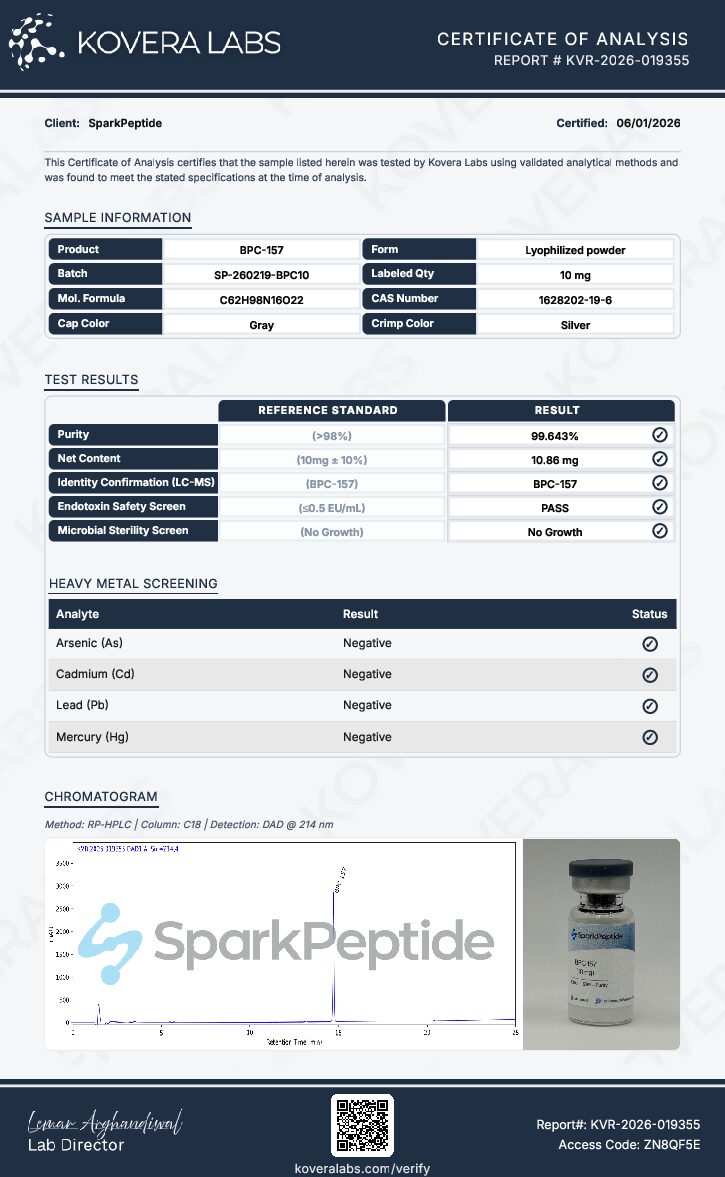
Every Spark Peptide product is supplied with batch-specific analytical documentation generated by independent third-party laboratories. These reports confirm compound identity, verify purity, and screen for contaminants. The documentation forms part of Spark Peptide’s 6X Safety Testing protocol, providing traceable analytical validation for laboratory research use.Certificate of Analysis (COA)
The Certificate of Analysis documents batch-specific analytical results for the manufacturing lot supplied to researchers. The report confirms peptide identity, purity verification, and safety screening parameters while providing traceability through lot numbers, analytical methods, and testing dates.HPLC Analysis Report
High-Performance Liquid Chromatography separates the peptide from potential impurities within the sample matrix. The resulting chromatogram allows researchers to quantify peptide purity by analyzing peak composition and relative abundance.Mass Spectrometry Report
Mass spectrometry confirms peptide identity by measuring the mass-to-charge ratio (m/z) of ionized molecules. The experimentally observed molecular mass is compared with the theoretical mass expected for the peptide sequence.Additional Safety Screening
In addition to HPLC and mass spectrometry verification, BPC-157 undergoes additional screening as part of Spark Peptide’s 6X Safety Testing protocol. These analyses include heavy metals testing for lead, mercury, arsenic, and cadmium, endotoxin testing using the LAL assay, and bacterial contamination analysis. Complete analytical reports are available upon request or through the Spark Peptide Tests & Safety page.Why 6X Safety Testing Matters for Your Research
While most suppliers verify purity alone, every SparkPeptide batch passes six independent quality and safety screenings before reaching your laboratory.| # | Test | What It Confirms |
| 1 | HPLC Purity Analysis | Peptide purity at 99.9%+ via reverse-phase chromatography |
| 2 | Mass Spectrometry | Correct molecular identity (observed vs. expected mass) |
| 3 | Heavy Metal Screening | Below detectable limits for lead, mercury, arsenic, cadmium |
| 4 | Endotoxin Testing | Bacterial endotoxin levels within safe research thresholds (LAL assay) |
| 5 | Bacterial Contamination | No microbial growth detected in culture testing |
| 6 | Solubility & Stability | Proper reconstitution behavior and structural integrity confirmed |
Legal Disclaimer
For Laboratory Research Use Only. All products sold by Spark Peptide are strictly intended for laboratory research use only. These materials are not for human consumption and are not intended for medical, veterinary, diagnostic, or household use of any kind. Spark Peptide operates solely as a research chemical supplier. We are not a compounding pharmacy and do not operate as a compounding facility as defined under Section 503A of the Federal Food, Drug, and Cosmetic Act. Additionally, Spark Peptide is not registered as an outsourcing facility under Section 503B of the Act. By purchasing from our site, you agree to use our products exclusively for lawful laboratory research purposes. Any misuse is strictly prohibited.Product FAQ for Researchers
What purity level does Spark Peptide’s BPC-157 peptide achieve?
Spark Peptide supplies BPC-157 peptide with 99.9%+ purity verified through high-performance liquid chromatography (HPLC). Each batch is further validated under Spark Peptide’s 6X Safety Testing protocol, which includes mass spectrometry identity confirmation, heavy metal screening, endotoxin testing, bacterial contamination analysis, and solubility and stability verification. These analytical controls support consistent quality and reproducibility for laboratory research applications.How should BPC-157 be reconstituted?
BPC-157 is typically reconstituted using sterile bacteriostatic water. Researchers should allow the vial to reach room temperature before adding the solvent slowly along the inner wall of the vial. Gentle swirling is recommended to dissolve the peptide, while vortexing or vigorous agitation should be avoided. Spark Peptide also offers a compatible preparation solvent, Bacteriostatic Water 10ml, for laboratory use.What is the recommended storage condition for BPC-157?
Lyophilized BPC-157 should be stored at -4°F (-20°C) in a sealed, desiccated vial protected from light and moisture. Under these conditions, the peptide typically maintains stability for up to 24 months. After reconstitution, the solution should be stored under refrigeration at 36–46°F (2–8°C) and used within standard laboratory stability timeframes.Does BPC-157 come with a Certificate of Analysis?
Yes. Every batch of BPC-157 supplied by Spark Peptide includes a batch-specific Certificate of Analysis (COA). The COA documents identity verification, purity testing results, and safety screening parameters for the manufacturing lot. Researchers can view or download the COA directly from the product page to confirm the analytical data associated with their batch.How is BPC-157 shipped to researchers ordering the peptide?
Spark Peptide ships BPC-157 in protective packaging designed to limit temperature fluctuations during transit, helping maintain stability for laboratories that buy BPC-157 peptide for experimental research. The compound is supplied in lyophilized form to maintain stability during shipping under standard delivery policies.What signaling pathways are commonly investigated with BPC-157?
Experimental studies involving the BPC-157 research peptide frequently examine nitric oxide signaling networks, endothelial regulatory pathways, and intracellular cascades such as PI3K/Akt and MAPK/ERK. These pathways are studied in experimental systems evaluating cellular migration, cytoskeletal organization, and extracellular matrix signaling in preclinical laboratory models.| Property | Detail |
|---|---|
| Name | BPC-157 (stable gastric pentadecapeptide) |
| Full Name | Body Protection Compound-157 |
| Sequence | Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val |
| Molecular Formula | C62H98N16O22 |
| Molecular Weight | ~1.4 kDa (varies slightly by salt form; confirm via COA) |
| Formats Available | BPC-157 5mg lyophilized powder; BPC-157 10mg lyophilized powder; BPC-157 500mcg tablets |
| Purity | ≥99% per component, verified by lot-specific Certificates of Analysis (COAs) |
| Solubility | Lyophilized powder soluble in bacteriostatic water or appropriate research-grade buffers; tablet forms intended for controlled laboratory dissolution |
| Storage (Powder) | –20°C (–4°F), protected from light and moisture; long-term: –80°C to –20°C |
| Storage (Tablets) | Sealed at room temperature or 2–8°C (35.8°F – 46.4°F), protected from humidity |
| Batch | EP-250407-BPC10 |
| Supplier | Spark Peptide |